Volume 1, Issue 2
Article Type: Review Article
Sleep disturbances in aging: Clinical evaluation and treatment of common disorders
MI Ullah, MD, MPH1*; Sadeka Tamanna, MD, MPH1,2
1University of Mississippi Medical Center, USA.
2GV (Sonny) Montgomery VA Medical Center, 1500 E Woodrow Wilson Ave, USA.
*Corresponding author: Mohammad I Ullah
Professor of Medicine, University of Mississippi Medical
Center, 2500 N State St, Jackson, MS, 39216, USA.
Tel: 601-984-5660, Fax: 601-984-6870; Email ID: mullah@umc.edu
Received: Jul 26, 2025
Accepted: Aug 13, 2025
Published Online: Aug 19, 2025
Journal: Annals of Gerontology and Geriatrics
Copyright: Ullah MI et al. © All rights are reserved
Citation: Ullah MI, Tamanna S. Sleep disturbances in aging: Clinical evaluation and treatment of common disorders. Ann Gerontol Geriatr Res. 2025; 1(2): 1018.
Abstract
Aging is accompanied by alterations in sleep architecture including reduced deep sleep, increased fragmentation, and earlier circadian timing, which may progress to clinical disorders and worsen health outcomes in many older adults. This review outlines normal age‑related sleep changes and examines the prevalence, risk factors, and impacts of common sleep disorders including insomnia, obstructive and central sleep apnea, restless legs syndrome, periodic limb movements, REM behavior disorder, and circadian rhythm disorders. We discuss contributory influences including neurodegeneration, vascular injury, chronic illnesses, medications, and psychosocial factors, and describe diagnostic approaches from structured interviews and questionnaires to polysomnography and actigraphy. Finally, we evaluate both conventional therapies as well as new emerging treatment options for various sleep disorders. By identifying gaps in research and care, this review emphasizes the need for tailored, multidisciplinary strategies to optimize sleep health and support healthy aging.
Introduction
Sleep is a fundamental biological process that supports cognitive function, metabolic regulation, and overall health. In older adults, however, both the quantity and quality of sleep undergo notable changes, contributing to increased morbidity and diminished quality of life [1,2]. As populations age worldwide, understanding how normal age‑related alterations in sleep evolve into clinical sleep disorders becomes critical for developing targeted interventions and improving geriatric care.
Physiologically, aging is associated with reductions in slow wave (deep) sleep and greater fragmentation of Rapid Eye Movement (REM) sleep, along with a decline in total sleep time and sleep efficiency [3,4]. Circadian rhythms also shift earlier (“phase advance”), leading to earlier bedtimes and awakenings, while homeostatic sleep drive diminishes, resulting in increased daytime napping [5]. Neurobiological changes, such as degeneration of the suprachiasmatic nucleus and decreased melatonin secretion, further contribute to these alterations [2]. Although some sleep changes are expected with healthy aging, many older individuals experience sleep disturbances that meet criteria for clinical disorders. Insomnia affects up to 50% of community dwelling elders, often linked to comorbid medical conditions, polypharmacy, and psychosocial stressors [5]. Sleep‑disordered breathing, particularly obstructive sleep apnea, becomes more prevalent with age and is associated with cardiovascular and cognitive sequelae [5]. Other conditions, such as restless legs syndrome, periodic limb movements, and REM sleep behavior disorder, also disproportionately affect older adults and can herald neurodegenerative disease [2,3].
The purpose of this review is to delineate the continuum from normal, physiologically driven changes in sleep architecture during aging to overt sleep pathology, and to evaluate current and emerging interventions aimed at preserving sleep health in later life. We will first characterize normative sleep alterations in the elderly, then examine the epidemiology and pathophysiology of common sleep disorders and finally discuss diagnostic approaches and evidence‑based management strategies tailored to the aging population.
Normal changes in sleep architecture with aging
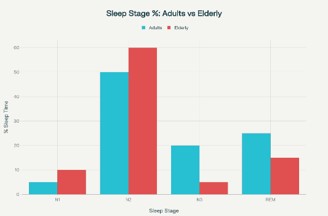
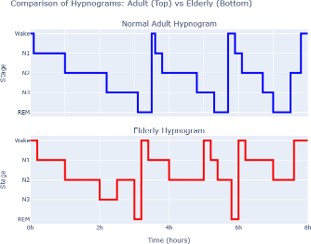
Aging is accompanied by distinct alterations in the quantity and quality of sleep (Figures 1 & 2). One of the most consistent findings is a decline in slow wave sleep, often referred to as deep sleep, with a corresponding increase in lighter sleep stages [3]. Older adults spend less time in stages of restorative slow wave sleep and more time in Stage 1 sleep, which is more easily disrupted by external stimuli. Rapid eye movement sleep also becomes more fragmented, leading to frequent brief arousals that reduce the continuity of the sleep cycle [4].
Circadian timing shifts earlier as people age, a phenomenon known as phase advance. This shift results in earlier bedtimes and awakenings even in the absence of external constraints [5]. The shift is believed to be driven by changes in the central circadian pacemaker, found in the suprachiasmatic nucleus of the hypothalamus. Reduced responsiveness to light cues and decreased amplitude of circadian rhythms further contribute to an earlier sleep phase [2].
Homeostatic sleep drive, which reflects the build‑up of sleep pressure during wakefulness, also diminishes with age. Older adults tend to experience a slower accumulation of sleep need, which can manifest as shorter nocturnal sleep episodes and increased propensity for daytime napping [5]. Although brief naps may partially compensate for reduced nocturnal sleep, they can further disrupt nighttime sleep continuity if taken too late in the day.
Sleep efficiency, defined as the ratio of total sleep time to time spent in bed, declines progressively in later life. Older individuals often require more time to fall asleep and awaken more frequently after sleep onset [3]. Increased sleep latency and greater wake after sleep onset contribute to a perception of poor sleep quality, even when total sleep time remains within an acceptable range [4].
Neurobiological changes accompany these alterations in sleep architecture. Degeneration of neurons in the suprachiasmatic nucleus reduces the precision of circadian signals. In parallel, the pineal gland’s production of melatonin declines, leading to lower circulating levels of this sleep promoting hormone [2]. Changes in hypothalamic and brainstem networks further impair the regulation of sleep–wake transitions, making sleep more fragile and prone to disruption.
Together, these normal changes in sleep architecture reflect an interaction of circadian, homeostatic, and neurobiological processes that evolve with age. While they do not constitute pathology on their own, they set the stage upon which clinical sleep disorders may develop in susceptible individuals.
Common sleep disorders in the elderly
Insomnia
Insomnia in older adults is characterized by persistent difficulty initiating or maintaining sleep, despite adequate opportunity, and is reported by up to half of community‑dwelling elders [6]. The overall prevalence of Insomnia ranges from 30% to 48% among elderly population [3,7]. Risk factors include chronic medical conditions, polypharmacy, mood disturbances, and environmental factors that fragment sleep [8]. Consequences extend beyond fatigue to include impaired cognition, mood disorders, increased risk of falls, and reduced quality of life.
Sleep disordered breathing
Sleep disordered breathing in older adults includes both obstructive and central events and becomes increasingly prevalent with age and is associated with substantial morbidity, including cardiovascular and cognitive consequences [9]. The odds of having obstructive sleep apnea are three times higher among people of >65 years old than overall risk of OSA in all ages [10]. Obstructive Sleep Apnea (OSA) results from recurrent collapse of the upper airway during sleep, leading to intermittent drops in blood oxygen levels and repeated arousals. In community samples, roughly one quarter of men and one in ten women over age sixty experience OSA of clinical significance [5,11]. Age related reductions in upper airway muscle tone, changes in fat distribution around the neck, and comorbid conditions such as obesity and chronic lung disease all increase OSA risk in the elderly [11]. Left untreated, OSA in older adults contributes to hypertension, stroke risk, and impairments in cognition and daytime function [5].
Central Sleep Apnea (CSA) is characterized by diminished or absent respiratory effort during sleep despite an open airway. While CSA is less common than OSA in the general population, its prevalence rises in elderly individuals, especially those with heart failure or neurological disease. In older patients with chronic heart failure, as many as one in four may exhibit CSA with Cheyne–Stokes breathing patterns [12]. Neurodegenerative disorders and strokes can also impair the brainstem respiratory centers, further increasing CSA risk in very late life [13]. CSA leads to sleep fragmentation, impaired ventilatory responses to carbon dioxide, and may exacerbate daytime fatigue and cognitive decline.
Because OSA and CSA often coexist in older adults, comprehensive assessment with polysomnography is essential to distinguish between the two and guide treatment choices. Continuous positive airway pressure remains first line for moderate to severe OSA, whereas adaptive servo‑ventilation or supplemental oxygen may be indicated for predominant CSA in selected patients, particularly those with heart failure [14].
Rapid eye movement sleep behavior disorder
Rapid Eye Movement Sleep Behavior Disorder (RBD) arises when the normal paralysis of voluntary muscles during REM sleep is lost, allowing patients to physically act out vivid dream content. Affected individuals commonly report punching, kicking, or shouting during sleep, which leads to frequent nocturnal arousals and fragmented sleep. This fragmentation reduces the restorative value of sleep and often results in excessive daytime sleepiness, impaired attention, and mood disturbances [15]. This disorder predominantly affects older adults with a prevalence rate of 0.4‑0.5% [16].
The impact of RBD extends to bed partners or spouses, who may be awakened by sudden movements or vocalizations. Concerns over injury to either the patient or the partner can lead to anxiety around bedtime, further disrupting sleep onset and continuity for both individuals. Partners may choose to sleep in separate rooms to avoid disturbance, which can erode intimacy and increase caregiver burden [17].
Longitudinal studies have demonstrated that idiopathic RBD often heralds neurodegenerative disease. Over half of patients with otherwise unexplained RBD develop an alpha synucleinopathy, most commonly Parkinson disease or Lewy body dementia, within ten years of RBD onset [18]. Pathologic studies have found widespread alpha synuclein deposits in brainstem nuclei responsible for REM atonia, suggesting that RBD reflects early neurodegeneration in these regions. Recognition of RBD thus provides a window for monitoring at risk individuals and exploring neuroprotective strategies before overt motor or cognitive symptoms appear.
Restless legs syndrome and periodic limb movement disorder
Restless Legs Syndrome (RLS) is a disorder characterized by an uncontrollable urge to move the legs, usually accompanied by uncomfortable sensations such as aching, crawling, tingling, or pulling. These symptoms typically occur during periods of rest or inactivity, especially in the evening or at night, and are temporarily relieved by movement. RLS often disrupts sleep and daytime functioning, and symptoms tend to worsen with age [19].
Periodic Limb Movement Disorder (PLMD) involves repetitive, involuntary limb movements during sleep, most commonly of the lower extremities. These movements can lead to sleep fragmentation and poor sleep quality. While PLMD often occurs in association with RLS, it can also present independently. Together they reduce total sleep time and sleep efficiency, contributing to excessive daytime sleepiness, impaired cognition, depressed mood, and reduced quality of life [20].
RLS is typically diagnosed with clinical history but PLMD is diagnosed with an in‑lab polysomnography [19]. Both RLS and PLMD increase in prevalence with age. Epidemiological studies estimate that up to 20 percent of adults older than sixty meet diagnostic criteria for restless legs syndrome, while periodic limb movements during sleep occur in nearly half of community‑dwelling [21].
Iron deficiency is a key risk factor for RLS. Low serum ferritin and reduced brain iron stores diminish dopamine synthesis in nigrostriatal pathways, exacerbating sensory and motor symptoms [21]. In elderly patients, comorbid conditions such as chronic kidney disease and nutritional deficits further increase the likelihood of restless legs syndrome.
Several medications commonly used in older adults can worsen these conditions. Dopamine antagonist agents such as certain antipsychotics and antiemetics may aggravate restless legs symptoms by blocking dopamine receptors. Antidepressant drugs, including selective serotonin reuptake inhibitors and tricyclic agents, have also been associated with symptomatic worsening [22]. Conversely, dopaminergic agonists can provide relief but require cautious dosing because of the risk of augmentation and impulse control disorders in later life.
Circadian rhythm sleep wake disorders
Advanced sleep phase disorder (ASPD)
It is the most commonly observed circadian rhythm sleep– wake disorders in older adults, in which the internal clock shifts earlier relative to the social and environmental schedule. Individuals with this condition begin to feel sleepy in the early evening, often before dinner, and wake up very early in the morning when most household members remain asleep. This misalignment leads to difficulty maintaining social routines, such as family meals or evening activities, and can contribute to social isolation and mood disturbances when patients must choose between staying awake uncomfortably or missing important events [5,19].
Early morning awakening may also reduce total sleep time if the individual cannot return to sleep after dawn. Over time, the combination of shortened nights and irregular social participation can erode quality of life by increasing daytime fatigue, impairing concentration, and exacerbating feelings of loneliness [2]. In addition, early evening sleepiness often conflicts with caregiver or spouse schedules, leading to mismatched bedtimes that can strain interpersonal relationships and diminish intimacy.
Neurological diseases such as stroke, Alzheimer disease, and Parkinson disease may further disrupt circadian organization in elderly patients. Cerebrovascular insults can damage pathways between the suprachiasmatic nucleus and other hypothalamic or brainstem nuclei, blunting the amplitude of circadian signals and altering phase timing [23]. In Alzheimer disease, degeneration of the molecular clockwork within the suprachiasmatic nucleus and decreased light responsiveness contribute to fragmented rhythms and sundowning phenomena [24,25]. Parkinson disease is likewise associated with reduced circadian amplitude and greater variability in sleep–wake cycles, which correlate with both motor and nonmotor symptom severity [2].
Irregular sleep-wake rhythm disorder (ISWRD)
This disorder is particularly prevalent in older adults with neurodegenerative diseases such as dementia and those residing in long‑term care facilities, often due to degeneration of the suprachiasmatic nucleus (the brain’s circadian pacemaker), decreased exposure to environmental time cues, and medical comorbidities. It is characterized by the absence of a clearly defined circadian sleep‑wake pattern, resulting in fragmented periods of sleep and wakefulness spread throughout the 24‑hour day rather than consolidated nighttime sleep and sustained daytime wakefulness. ISWRD leads to significant impairment in nocturnal sleep quality and increased daytime napping, which can further exacerbate cognitive and functional decline in this population [19,26].
Together, these influences can worsen natural age‑related phase advances, creating difficulties for older people, particularly those with neurological diseases, in keeping up with societal expectations. Recognition of circadian rhythm sleep–wake disorders is therefore essential for developing appropriate interventions that aim to realign internal clocks with daily activities.
Contributing factors to sleep pathology in aging
Neurological degeneration
Progressive neurodegenerative disorders undermine the integrity of circuits that regulate sleep and wakefulness. In Alzheimer disease, accumulation of amyloid plaques and neurofibrillary tangles in and around the suprachiasmatic nucleus weakens circadian signaling and leads to fragmented sleep– wake cycles and sundowning phenomena [25]. In Parkinson disease, degeneration of brainstem nuclei that normally generate rapid eye movement atonia and modulate arousal produces not only REM sleep behavior disorder but also reduced sleep efficiency, multiple nocturnal awakenings, and excessive daytime sleepiness [18]. Even in the absence of a formal diagnosis, age related declines in cholinergic and monoaminergic neurotransmission contribute to lighter, more disrupted sleep across the elderly population [27].
Cerebrovascular events such as stroke often damage pathways essential for sleep regulation. Up to half of acute stroke survivors experience new‑onset insomnia or daytime hypersomnia, depending on lesion location and extent. Infarcts in thalamic or brainstem regions may blunt sleep spindle generation and slow wave activity, whereas cortical strokes can disrupt frontal control over arousal thresholds. These changes prolong sleep latency, increase nocturnal awakenings, and contribute to daytime fatigue and cognitive slowing [28,29].
Traumatic Brain Injury (TBI) in older adults likewise predisposes to a spectrum of sleep disorders. In a systematic review, traumatic injury was associated with insomnia, hypersomnia, and sleep‑related breathing disorders in up to 70 percent of patients [30]. Diffuse axonal injury and focal contusions can impair hypothalamic and brain‑stem networks that coordinate sleep architecture, leading to reduced slow wave sleep, circadian disruption, and persistent daytime somnolence.
Other neurological conditions including multiple sclerosis, epilepsy, and peripheral neuropathies also impair sleep through diverse mechanisms. Demyelinating lesions in multiple sclerosis can damage central circadian pacemakers and reduce melatonin secretion, producing both insomnia and excessive daytime [31]. Nocturnal seizure activity in epilepsy and neuropathic pain syndromes fragment sleep continuity and diminish restorative sleep stages, further eroding quality of life.
Across these disorders, the common thread is disruption of the neuroanatomical and neurochemical substrates governing sleep stability, timing, and depth. Recognition of these links informs both diagnostic evaluations, highlighting the need for targeted neuroimaging or polysomnography, and therapeutic strategies that address the underlying neurological insult alongside standard sleep interventions.
Chronic illnesses and medications
Multiple chronic conditions common in late life, such as heart failure, chronic obstructive pulmonary disease, arthritis, and gastroesophageal reflux produce nocturnal symptoms that interrupt sleep [6]. Pain, dyspnea, and nocturia each can trigger awakenings and reduce sleep continuity. Moreover, polypharmacy increases the risk of sleep‑disrupting side effects. Diuretics may provoke nocturia, beta blockers can suppress melatonin secretion, and corticosteroids often cause insomnia and restlessness [3]. Awareness of medication timing and selection of sleep‑friendly agents are essential in geriatric practice.
Psychological factors
Depression and anxiety both rise in prevalence with age and exert powerful effects on sleep. Depressive symptoms are associated with early morning awakening and nonrestorative sleep, while anxiety often prolongs sleep latency through rumination and hyperarousal [8]. Feelings of loneliness and bereavement amplify these effects: perceived social isolation correlates with increased sleep fragmentation and poorer subjective sleep quality in elderly populations [32]. Addressing mood disorders and social support therefore plays a dual role in improving mental health and sleep.
Social and environmental aspects
Changes in daily routines and living situations can disrupt sleep–wake rhythms. Retirement often reduces daytime structure and exposure to daytime light, weakening circadian entrainment [5]. Lack of opportunities for daytime activities, especially in some nursing home settings, may encourage daytime napping and further erode nocturnal sleep pressure [33]. Environmental factors such as noise and uncomfortable bedding may worsen sleep quality and merit targeted interventions to restore healthy sleep habits.
Diagnostic approches
Accurate diagnosis of sleep disturbances in elderly individuals relies on a combination of clinical assessment tools and objective measurements. A structured approach helps distinguish between normal age‑related changes and pathological conditions, guiding appropriate intervention [34].
Clinical interview and sleep history
A thorough clinical interview remains the cornerstone of sleep evaluation. Key elements include bedtime and wake time patterns, sleep latency, number and duration of nocturnal awakenings, daytime sleepiness, and napping behavior [35]. Inquiry into lifestyle factors, caffeine and alcohol use, medical comorbidities, and medication timing helps identify reversible contributors. Collateral history from bed partners can elucidate nocturnal behaviors such as apneas or limb movements that the patient may not recall.
Sleep questionnaires and diaries
Validated instruments quantify subjective sleep quality and daytime functioning. The Pittsburgh Sleep Quality Index assesses multiple domains of sleep over the prior month, yielding a global score that distinguishes good from poor sleepers [36]. The Epworth Sleepiness Scale measures propensity to doze in routine situations and guides evaluation of excessive daytime sleepiness [37]. A two‑week sleep diary provides daily data on sleep–wake timing, enabling visualization of circadian patterns and response to interventions [38].
Polysomnography
Overnight in‑laboratory polysomnography is the gold standard for diagnosing many sleep disorders. It records electroencephalogram, electrooculogram, electromyogram, airflow, respiratory effort, oxygen saturation, and limb movements [39]. Polysomnography quantifies sleep architecture changes, identifies apneas and hypopneas for sleep disordered breathing, and detects periodic limb movements. In elderly patients, home portable monitoring may be used for screening obstructive sleep apnea, but full polysomnography is recommended when central apneas, REM behavior disorder, or complex comorbidities are suspected [40].
Actigraphy
Wrist actigraphy provides a noninvasive method to estimate sleep–wake patterns over multiple days or weeks. It measures movement to infer sleep efficiency, sleep duration, and circadian phase [41]. Actigraphy is especially useful for identifying circadian rhythm disorders, quantifying napping behavior, and monitoring response to interventions such as light therapy. While less precise than polysomnography for sleep staging, it offers ecological validity in the patient’s home environment [42].
Management of insomnia
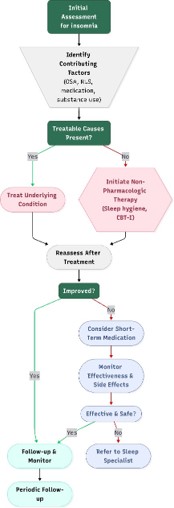
Management of insomnia in elderly patients centers on identifying and addressing underlying medical, psychiatric, or environmental contributors through careful assessment (Figure 3).
Non-pharmacologic interventions
Sleep hygiene education
Sleep hygiene education involves promoting behavioral and environmental strategies to improve sleep quality in older adults. Common recommendations include maintaining a consistent sleep schedule, optimizing the sleep environment, avoiding caffeine or vigorous exercise in the evening, and reducing exposure to screens before bedtime. Intervention studies using tools such as the Pittsburgh Sleep Quality Index (PSQI) have shown that sleep hygiene education can significantly improve self‑reported sleep quality among older adults. As a cost-effective and low‑risk intervention, sleep hygiene education is recommended as a first‑line strategy for sleep management in the elderly [43].
Cognitive behavioral therapy for insomnia
Cognitive behavioral Therapy for Insomnia (CBT‑I) is considered the gold‑standard non‑pharmacologic treatment for insomnia in adults, including the elderly. It targets maladaptive thoughts, behaviors, and emotions that impede sleep. Studies demonstrate that CBT‑I significantly improves sleep onset, wake after sleep onset, total sleep time, and sleep quality in older adults—even in those with comorbidities such as chronic pain. Importantly, CBT‑I’s benefits are durable, with evidence supporting its long‑term effectiveness and minimal side effects [44,45].
Bright light therapy
Bright Light Therapy (BLT) consists of controlled exposure to high‑intensity light to regulate circadian rhythms and improve sleep architecture in aging populations. Clinical trials in institutionalized older adults demonstrate that BLT can enhance circadian rhythm regularity, increase sleep efficiency, reduce nocturnal awakenings, and even improve cognitive function. BLT is generally well‑tolerated and may be particularly useful in patients with circadian rhythm disorders or diminished exposure to natural daylight [46,47].
Exercise and mindfulness
Physical activity, especially moderate‑intensity aerobic exercise, and mindfulness‑based practices (e.g., meditation, gentle yoga) have been shown to benefit sleep quality in older adults. Exercise should be scheduled earlier in the day to prevent overstimulation near bedtime. Mindfulness and meditation help manage stress and intrusive thoughts, which are common barriers to sleep in aging, and are accessible interventions safe for a wide range of functional abilities [48,49].
Pharmacologic treatments
Pharmacologic treatments for insomnia in older adults (Table 1) must be selected with caution due to increased sensitivity to medications, altered pharmacokinetics, and heightened risk of adverse effects such as falls, delirium, and cognitive impairment. While non‑pharmacologic interventions remain first‑line, several medication classes may be considered for short‑term or adjunctive use when behavioral therapy is insufficient [50,51].
Melatonin and melatonin receptor agonists like ramelteon are among the safest options for elderly patients. These agents help improve sleep onset and are particularly useful for circadian rhythm disorders or insomnia associated with age‑related declines in endogenous melatonin. Melatonin is generally well tolerated, with few side effects such as headache, dizziness, or vivid dreams. Ramelteon, a prescription alternative, offers longer‑lasting effects and similarly low risk for dependence or next‑day sedation. However, caution is advised in patients with autoimmunity or hepatic impairment.
Low-dose doxepin (3‑6 mg) is another well‑supported option, especially effective for sleep maintenance insomnia. At such low doses, it has minimal anticholinergic effects, though patients may experience dry mouth or mild drowsiness. Doxepin should be avoided in individuals with glaucoma, urinary retention, or significant cardiovascular disorders.
Dual Orexin Receptor Antagonists (DORAs) such as suvorexant and lemborexant are newer agents demonstrated to improve both sleep onset and maintenance without causing significant cognitive or respiratory depression. These medications are generally well‑tolerated in older adults but may be associated with unusual side effects such as sleep paralysis, abnormal dreams, or complex sleep‑related behaviors. DORAs should be avoided in individuals with narcolepsy and used cautiously in those taking CYP3A inhibitors or with untreated sleep apnea or COPD.
Non-benzodiazepine GABA agonists, often called “Z‑drugs” (e.g., zolpidem, eszopiclone), are effective short‑term treatments for sleep initiation or maintenance but require caution in the elderly due to their association with next‑day sedation, confusion, dizziness, risk of falls, and complex sleep behaviors. These agents are metabolized more slowly in older adults and may accumulate, increasing the risk for cognitive impairment and delirium. They should be used only when safer alternatives are ineffective and for the shortest duration possible.
Sedating antidepressants, such as trazodone and mirtazapine, are sometimes used off‑label to treat insomnia, particularly when comorbid depression or anxiety is present. Although they may facilitate sleep, their anticholinergic and hypotensive effects increase fall risk. Trazodone is associated with dizziness and orthostatic hypotension, while mirtazapine can cause significant weight gain, daytime sedation, or confusion.
Benzodiazepines such as temazepam are no longer recommended for routine use in elderly patients with insomnia. Despite their short‑term efficacy, these medications significantly increase risk of falls, confusion, respiratory depression, and dependence. They are particularly hazardous in individuals with a history of substance use disorder, cognitive impairment, or respiratory disease.
The first‑generation antihistamines (e.g., diphenhydramine) should be avoided in the elderly due to strong anticholinergic effects which may precipitate delirium, urinary retention, blurred vision, or constipation. These medications, often found in over‑the‑counter sleep aids, are widely discouraged by geriatric guidelines such as the Beers Criteria [52].
In all cases, pharmacologic therapy in older adults should be used at the lowest effective dose, for the shortest duration, with periodic reassessment and an accompanying non‑pharmacologic strategy. Continual monitoring for side effects and functional outcomes is essential to ensure safety and efficacy.
Role of polypharmacy in sleep disturbance in elderly
Medications and polypharmacy frequently contribute to sleep disturbances in elderly individuals, manifesting as both insomnia and hypersomnia. Older adults often take multiple medications for chronic conditions, increasing the risk of adverse effects that impair sleep quality. Common offenders include diuretics causing nocturia with frequent nighttime awakenings; beta‑blockers and corticosteroids inducing insomnia and vivid dreams; and certain antidepressants (e.g., SSRIs) that may provoke insomnia or fragmented sleep. Conversely, sedative medications intended to aid sleep can result in daytime drowsiness and poor sleep architecture, potentially worsening hypersomnia symptoms. Drug interactions and anticholinergic burden can also lead to cognitive impairment, confusion, and delirium, further disrupting normal sleep‑wake patterns [53,54].
Management involves regular medication review with an emphasis on deprescribing when possible, especially of potentially inappropriate medications that adversely affect sleep. Clinicians should seek to minimize polypharmacy by prioritizing essential drugs, considering drug side effect profiles, and adjusting dosing schedules to reduce nighttime sleep disruption. Behavioral strategies and non‑pharmacologic treatments should be maximized to avoid introducing additional sedative medications. Collaboration with pharmacists and use of tools such as the Beers Criteria can guide safer prescribing [52]. Ongoing monitoring for sleep quality changes after medication adjustments is critical, as is educating patients and caregivers about the impact of medications on sleep and daytime functioning.
Table 1: Different medications to treat insomnia in elderly.
| Drug Class | Safety/efficacy | Main side effects | Cautions/contraindications |
|---|---|---|---|
| Melatonin/Ramelteon | Good efficacy, high safety | Vivid dreams, headache, dizziness | Autoimmune disease, hepatic impairment |
| Doxepin (low-dose) | Modest efficacy, good safety | Dry mouth, sedation | Glaucoma, urinary retention, cardiac arrhythmia |
| DORAs (Suvorexant, Lemborexant) | Good efficacy, generally safe | Somnolence, parasomnias, abnormal dreams | Narcolepsy, CYP3A inhibitors, severe sleep apnea |
| Z-drugs (Zolpidem, Eszopiclone) | Effective, but higher risks in elderly | Falls, confusion, next‑day impairment | Dementia, respiratory disease, high fall risk |
| Sedating antidepressants (Trazodone) | Useful with depression/anxiety, but side effects | Hypotension, dizziness, confusion | Cardiovascular disease, increased fall risk |
| Benzodiazepines | Effective, but not recommended | Delirium, falls, dependence | Cognitive impairment, COPD, substance use, falls |
| Antihistamines | Not recommended | Confusion, delirium, anticholinergic effects | All elderly (avoid use entirely) |
Management of sleep disordered breathing
Obstructive sleep apnea
The management of OSA in the elderly follows similar principles as in younger adults, but must be individualized due to age‑related comorbidities, changes in sleep architecture, and challenges with treatment adherence.
Continuous Positive Airway Pressure (CPAP) remains the gold standard treatment for moderate to severe OSA in elderly patients and is associated with improvements in sleep quality, daytime alertness, mood, cognition, and prevention of cardiovascular and cerebrovascular events—particularly in those with severe disease [55]. However, compliance may decline with age, especially in individuals over 80 years or with cognitive impairment; strategies such as therapeutic education, mask adaptation, and telemonitoring are crucial to optimize adherence [9].
Hypoglossal Nerve Stimulation (HNS) is an implantable device therapy for OSA that has become an important alternative for elderly patients who are intolerant of CPAP or other conventional therapies. HNS works by delivering electrical stimulation to the hypoglossal nerve, which activates upper airway muscles to maintain airway patency during sleep, thus reducing apneic events. In elderly patients, HNS is shown to be safe and effective, with high adherence rates and significant improvements in sleep quality and daytime functioning, especially when CPAP use is limited by factors such as reduced dexterity, dental issues, or intolerance of mask‑based therapy [56]. However, challenges may include managing comorbidities, polypharmacy, and cognitive impairment that can affect device use and follow‑up, making patient selection and support critical for optimal outcomes [9].
Other Interventions where CPAP is not tolerated or in milder disease include custom‑fit oral appliances (mandibular advancement devices), positional therapy to reduce supine sleep, and adjunctive measures such as weight loss, smoking cessation, and reduction of sedating medications [9,55]. Surgical interventions are rarely used in the elderly but may be considered in select cases.
Tirzepatide (Zepbound®), a dual GLP‑1/GIP receptor agonist originally developed for type 2 diabetes and obesity, has emerged as a promising option for weight reduction, a key modifiable risk factor in OSA. In elderly patients with obesityrelated OSA who are unable or unwilling to use primary treatments like CPAP, tirzepatide may assist by promoting significant weight loss, which has been shown to reduce the severity of OSA events and improve associated comorbidities [9]. However, practitioners must exercise caution as elderly individuals are more vulnerable to gastrointestinal side effects (such as nausea, vomiting, and diarrhea), volume depletion, hypoglycemia (especially when combined with other antidiabetic agents), sarcopenia, and potentially increased risk of gallbladder or renal problems. Furthermore, while weight loss may improve OSA severity, tirzepatide is not a direct therapy for upper airway collapsibility and should not replace established OSA treatments like CPAP or oral appliances. Overall, tirzepatide may be considered as an adjunct for meticulously selected elderly patients with obesity‑related OSA but requires careful monitoring for tolerability and potential adverse events, and regular reassessment of OSA management needs.
Central sleep apnea
Central Sleep Apnea (CSA) in elderly patients is frequently associated with medical conditions such as heart failure, stroke, renal insufficiency, or the use of opioids. The management of CSA begins with identifying and treating any underlying causes, as optimization of these conditions can often lead to significant improvement or even resolution of the apnea events. For example, effective treatment of congestive heart failure or discontinuation of offending medications may substantially reduce central apneas [19].
Positive Airway Pressure (PAP) therapies are central to symptomatic management. Continuous Positive Airway Pressure (CPAP) is commonly employed first, as it helps maintain airway patency and improves ventilation. However, CPAP’s effectiveness may be limited in pure CSA or cases complicated by Cheyne‑Stokes respiration, resulting in incomplete resolution of apnea events. Adaptive Servo‑Ventilation (ASV), which provides dynamic ventilatory support by adjusting inspiratory pressure on a breath‑by‑breath basis, has demonstrated notable efficacy in many patients with CSA, especially those with idiopathic CSA or CSA associated with heart failure without reduced ejection fraction. Despite these benefits, ASV is contraindicated in patients with reduced left ventricular ejection fraction (less than 45%) due to evidence linking its use in this population with increased mortality [57].
Bilevel Positive Airway Pressure (BPAP), especially with a Spontaneous/Timed (ST) mode, is an important treatment modality for Central Sleep Apnea (CSA), particularly in elderly patients who may exhibit hypoventilation in addition to central apneas. Unlike CPAP, BPAP delivers two pressure levels: a higher Inspiratory Positive Airway Pressure (IPAP) and a lower Expiratory Positive Airway Pressure (EPAP), providing pressure support that assists ventilation. The ST mode incorporates a backup respiratory rate that delivers mandatory breaths if the patient’s spontaneous respiratory effort falls below a set threshold, thus preventing prolonged apnea or hypopnea episodes due to diminished central respiratory drive.
BPAP‑ST is particularly useful in patients with CSA combined with hypoventilation syndromes, such as those with heart failure, neuromuscular disorders, or opioid‑induced respiratory depression, where augmenting ventilation through pressure support and timed breaths can stabilize breathing patterns during sleep [58,59]. During BPAP titration in a sleep laboratory, settings including IPAP, EPAP, backup rate, and inspiratory time are adjusted to maintain upper airway patency, optimize ventilation, and minimize central apneas. However, caution is warranted as BPAP without careful titration may sometimes worsen central apneas or induce periodic breathing patterns due to overcompensation, particularly if pressure settings are too aggressive or patient‑ventilator synchrony is poor [60], Patient‑device asynchrony and air leaks can contribute to discomfort and residual sleep disruption, emphasizing the need for polysomnography‑guided titration and follow‑up monitoring to ensure efficacy and tolerability.
A relatively novel therapeutic option for CSA is implantable phrenic nerve stimulation. This approach delivers electrical stimulation to the phrenic nerve, activating the diaphragm and thus restoring respiratory effort during apneic episodes. Clinical studies have demonstrated improvements in apnea‑hypopnea indices and patient‑reported sleep quality with this technique [61,62]. The therapy demands specialized centers for implantation and follow‑up, and thus, it is not available in many hospitals or sleep disorder clinics. But it can be used as an alternative form of treatment for CSA when appropriate.
Management of REM behavior disorder
Rapid Eye Movement Sleep Behavior Disorder (RBD) is most commonly seen in elderly populations. Management in this population requires an emphasis on both symptom control and safety, and should also include screening for associated neurodegenerative conditions, given the strong association between RBD and synucleinopathies such as Parkinson’s disease, Lewy body dementia, and multiple system atrophy [63].
The mainstay of RBD treatment is environmental modification to prevent injury. Elderly patients and their bed partners should be advised to remove hazardous objects from the sleep environment, pad corners, place the mattress on the floor if needed, and consider protective barriers or bed rails. For patients with severe, uncontrolled RBD, sleeping separately from their partner is advised, or at the very least, placing a pillow as a barrier between them. Such measures are essential as violent dream‑enactment can result in substantial harm, especially in older adults with comorbidities and impaired mobility [64].
Pharmacotherapy is indicated for persistent or severe cases. Clonazepam, at doses ranging from 0.25–1mg at bedtime, has traditionally been first‑line therapy and is effective in reducing RBD events in approximately 80–90% of patients [18]. However, caution is crucial in the elderly due to the risk of sedation, cognitive impairment, gait instability, and falls, particularly in those with dementia or other frailties. Melatonin (3–15 mg at bedtime) may also be used as an initial agent in this age group due to its favorable safety profile and few adverse effects. It is especially useful for those intolerant to clonazepam, and may improve both RBD symptoms and sleep quality; in some patients, a slow titration of the dose (starting at 3 mg and increasing by 3‑mg increments to 15 mg) may be necessary to balance efficacy and tolerability [64].
If monotherapy is insufficient, some cases may benefit from a combination of melatonin and reduced‑dose clonazepam, or, less commonly, other agents such as pramipexole or rivastigmine, though evidence for these alternatives is limited. Importantly, clinicians should regularly reassess for neurodegenerative symptoms, as a large proportion of patients with idiopathic RBD eventually develop Parkinsonism or dementia.
Management of RLS and PLMD
Restless Legs Syndrome (RLS) and Periodic Limb Movement Disorder (PLMD) are common sleep‑related movement disorders in elderly populations, frequently leading to significant sleep disruption, distress, and impaired daytime functioning. Management in older adults requires a comprehensive and individualized approach that takes into account age‑related physiological changes, comorbid conditions, polypharmacy, and potential treatment side effects.
The initial approach to managing RLS should focus on identifying and modifying contributing factors, such as alcohol and caffeine intake, as well as medications with antihistaminergic, serotonergic, or antidopaminergic effects. Additionally, untreated obstructive sleep apnea should be addressed. Since iron deficiency can cause RLS, all patients with clinically significant RLS should have serum iron studies including ferritin and transferrin saturation. Iron supplementation, either oral or intravenous, is recommended if the ferritin is ≤75 ng/mL or transferrin saturation is <20%, with intravenous preparations preferred in certain cases for better efficacy and tolerability [65].
If iron deficiency and other contributing factors have been addressed or ruled out, pharmacological treatment may need to be initiated if symptoms are still not resolved. The American Academy of Sleep Medicine (AASM) 2025 clinical practice guideline recommends gabapentin, gabapentin enacarbil and pregabalin as first‑line agents due to their efficacy in reducing sensory symptoms and improving sleep with a lower risk of augmentation. These gabapentinoids should be initiated at lower doses in older adults (e.g., gabapentin at 100 mg daily and pregabalin at 50 mg daily) with slow titration to balance efficacy and tolerability. Dopaminergic agents, such as pramipexole, are no longer favored due to a higher risk of augmentation and exacerbation of symptoms, especially with long‑term use, and are recommended against as first‑line therapy in older populations [65].
Opioids are considered as secondor third‑line treatments, particularly in refractory cases or when other agents are contraindicated or not tolerated. Their use mandates caution in elderly patients due to risks of sedation, cognitive impairment, falls, and dependency. Additionally, the guideline suggests against the routine use of benzodiazepines, clonazepam, and other sedative‑hypnotics due to limited evidence and risk profile in this demographic.
PLMD, which often coexists with or is secondary to RLS, may also benefit from treatment targeting the underlying RLS. Isolated PLMD without significant RLS symptoms generally does not require pharmacologic treatment unless it causes marked sleep fragmentation or daytime impairment [65].
In all treatment decisions, the elderly patient’s comorbidities, renal function, cognitive status, and potential drug interactions must be carefully considered to minimize side effects and optimize outcomes. Regular monitoring and dose adjustments are essential. For those with renal impairment, dose modification of gabapentinoids and other treatments is important due to altered drug clearance in older adults.
Management of circadian rhythm sleep disorder
Advanced Sleep Phase Disorder (ASPD)
The primary treatment goal is to delay the circadian phase to achieve a more conventional sleep‑wake schedule and enhance quality of life.
1. Bright light therapy (BLT)
Bright light therapy is the cornerstone intervention for ASPD in the elderly. Scheduled exposure to bright artificial light (typically 3,000–10,000 lux), administered in the early evening, can delay the endogenous circadian rhythm and shift both bedtime and wake time later [46]. Light therapy sessions usually last 30–60minutes and must continue daily for several weeks to achieve clinically meaningful results. For optimal impact, the light source should be at or positioned slightly above eye level (about 30‑90 cm away), and exposure should be indirect, with patients keeping their eyes open without looking directly at the light. Exposure to morning light could reinforce the early circadian phase and reduce the effectiveness of evening BLT. Thus, the patients should avoid bright morning light by remaining in dim environments upon awakening or using blackout curtains.
2. Chronobiotic medications
Exogenous melatonin is generally used to advance sleep, but some data suggest that very low doses (0.5–1mg) taken in the early morning could contribute to a phase delay when combined with evening BLT, though evidence is inconclusive and this approach is not established as a first‑line treatment [46,66] The role of melatonin receptor agonists such as ramelteon in ASPD remains limited and is best reserved for cases where nonpharmacologic interventions are insufficient.
3. Behavioral and environmental interventions
• Behavioral strategies may be used as a complement to light therapy in treating ASPD.
• Older adults should maintain consistent (but progressively later) sleep and wake times to facilitate phase shifting.
• Increasing stimulating activities (social or light physical exercise) in the early evening can reinforce later bedtimes.
• Minimizing or eliminating daytime napping helps consolidate sleep.
• Environmental controls, such as using bright lighting in the early evening and dim lighting in the morning, reinforce circadian realignment [46].
4. Monitoring and special considerations
Close follow‑up is important to monitor effectiveness, maintain adherence, and address any safety concerns, given the elevated risk of falls and confusion in elderly individuals. Polypharmacy and sedating medications should be avoided to minimize risk of cognitive and functional impairment.
5. Adjunct therapies
CBT‑I (Cognitive Behavioral Therapy for Insomnia) is a valuable adjunct for persistent insomnia symptoms, particularly for difficulties with sleep maintenance, and can be used in combination with BLT and behavioral adjustments.
Irregular sleep-wake rhythm disorder
Management of irregular sleep wake rhythm disorder (ISWRD) in elderly focuses on consolidating nighttime sleep and promoting daytime alertness through non‑pharmacological interventions. The most important strategies involve structured exposure to bright light during the day, especially in the morning and early afternoon, which helps to synchronize the circadian system and decrease daytime napping [26]. Incorporating morning outdoor time with light exercise, such as walking, is an ideal addition to daily home or institutional schedules. To ensure sufficient light reaches the eyes, it’s best to avoid wearing sunglasses. Enhancing physical and social activity during daytime hours and maintaining a consistent daily schedule for sleep, meals, and activities are also essential for strengthening circadian time cues [67]. A favorable sleep environment, minimizing light and noise at night, further supports nocturnal sleep consolidation. Pharmacological therapies, such as melatonin or hypnotics, generally provide limited benefits and may carry risks in the elderly; thus, treatment is best centered on multicomponent behavioral approaches tailored to individual needs.
Conclusions
Sleep disorders are common among elderly individuals, driven by age‑related changes, comorbidities, and environmental factors. Effective management requires a personalized approach that balances safety with symptom control. Sleep onset and sleep maintenance insomnia are the most common sleep disorders in this population. Insomnia treatment prioritizes cognitive behavioral therapy as first‑line, supplemented cautiously by pharmacologic options. For sleep apnea, therapies including CPAP, bilevel PAP, and emerging neural stimulation devices offer benefit but demand careful patient selection and monitoring. Beyond OSA and insomnia, sleep disorders in the elderly are driven by age‑related changes, comorbidities, and environmental factors, necessitating a personalized approach that balances safety with symptom control. Overall, interdisciplinary care, ongoing assessment, and caregiver support are essential to improve sleep quality and quality of life in older adults. Continued research is needed to refine strategies for this growing population.
References
- Grandner MA. Sleep duration across the lifespan: implications for health. Sleep Med Rev. 2012; 16: 199–201.
- Mander BA, Winer JR, Walker MP. Sleep and Human Aging. Neuron. 2017; 94: 19–36.
- Crowley K. Sleep and sleep disorders in older adults. Neuropsychol Rev. 2011; 21: 41–53.
- Ohayon MM, Carskadon MA, Guilleminault C, Vitiello MV. Metaanalysis of quantitative sleep parameters from childhood to old age in healthy individuals: developing normative sleep values across the human lifespan. Sleep. 2004; 27: 1255–73.
- Ancoli‑Israel S, Ayalon L, Salzman C. Sleep in the elderly: normal variations and common sleep disorders. Harv Rev Psychiatry. 2008; 16: 279–86.
- Foley D, Ancoli‑Israel S, Britz P, Walsh J. Sleep disturbances and chronic disease in older adults: Results of the 2003 National Sleep Foundation Sleep in America Survey. J Psychosom Res. 2004; 56: 497–502.
- Ohayon MM. Epidemiology of insomnia: what we know and what we still need to learn. Sleep Med Rev. 2002; 6: 97–111.
- Morin CM, Benca R. Chronic insomnia. Lancet Lond Engl. 2012; 379: 1129–41.
- Joskin A, Bruyneel M. Challenges in Obstructive Sleep Apnea Management in Elderly Patients. J Clin Med. 2024; 13: 7718.
- Ernst G, Mariani J, Blanco M, Finn B, Salvado A, Borsini E. Increase in the frequency of obstructive sleep apnea in elderly people. Sleep Sci Sao Paulo Braz. 2019; 12: 222–6.
- Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, Hla KM. Increased prevalence of sleep‑disordered breathing in adults. Am J Epidemiol. 2013; 177: 1006–14.
- Lanfranchi PA, Somers VK. Sleep‑disordered breathing in heart failure: characteristics and implications. Respir Physiol Neurobiol. 2003; 136: 153–65.
- Ishikawa O, Oks M. Central Sleep Apnea. Clin Geriatr Med. 2021; 37: 469–81.
- Javaheri S. Central sleep apnea in congestive heart failure: prevalence, mechanisms, impact, and therapeutic options. Semin Respir Crit Care Med. 2005; 26: 44–55.
- Postuma RB, Arnulf I, Hogl B, Iranzo A, Miyamoto T, Dauvilliers Y, et al. A single‑question screen for rapid eye movement sleep behavior disorder: a multicenter validation study. Mov Disord Off J Mov Disord Soc. 2012; 27: 913–6.
- Trotti LM. REM sleep behaviour disorder in older individuals: epidemiology, pathophysiology and management. Drugs Aging. 2010; 27: 457–70.
- Lam SP, Wong CCY, Li SX, Zhang JH, Chan JW, Zhou JY, et al. Caring burden of REM sleep behavior disorder – spouses’ health and marital relationship. Sleep Med. 2016; 24: 40–3.
- St Louis EK, Boeve AR, Boeve BF. REM Sleep Behavior Disorder in Parkinson’s Disease and Other Synucleinopathies. Mov Disord. 2017; 32: 645–58.
- AASM. International classification of sleep disorders (ICSD‑3TR). Darien, IL: American Academy of Sleep Medicine; 2023.
- Mungan S, Ozcan M, Orhan G, Titiz A, Arli B, Koseoglu S, et al. Clinical and polysomnographic features of patients with Restless Legs Syndrome. JPMA J Pak Med Assoc. 2016; 66: 568–73.
- Allen RP, Picchietti D, Hening WA, Trenkwalder C, Walters AS, Montplaisi J, et al. Restless legs syndrome: diagnostic criteria, special considerations, and epidemiology. A report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Med. 2003; 4: 101–19.
- Castillo‑Álvarez F, Marzo‑Sola ME. Restless legs syndrome. Pathophysiology, diagnosis and treatment. Med Clin (Barc). 2025; 164: 84–90.
- Duffy JF, Czeisler CA. Age‑related change in the relationship between circadian period, circadian phase, and diurnal preference in humans. Neurosci Lett. 2002; 318: 117–20.
- Satlin A, Volicer L, Stopa EG, Harper D. Circadian locomotor activity and core‑body temperature rhythms in Alzheimer’s disease. Neurobiol Aging. 1995; 16: 765–71.
- Volicer L, Harper DG, Manning BC, Goldstein R, Satlin A. Sundowning and circadian rhythms in Alzheimer’s disease. Am J Psychiatry. 2001; 158: 704–11.
- Kolberg E, Pallesen S, Hjetland GJ, Nordhus IH, Flo‑Groeneboom E. The Effect of Bright Light Treatment on Rest–Activity Rhythms in People with Dementia: A 24‑Week Cluster Randomized Controlled Trial. Clocks Sleep. 2021; 3: 449–64.
- Bliwise DL. Sleep in normal aging and dementia. Sleep. 1993; 16: 40–81.
- Baillieul S, Dekkers M, Brill AK, Schmidt MH, Detante O, Pépin JL, et al. Sleep apnoea and ischaemic stroke: current knowledge and future directions. Lancet Neurol. 2022; 21: 78–88.
- Javaheri S, Barbe F, Campos‑Rodriguez F, Dempsey JA, Khayat R, Javaheri S, et al. Sleep Apnea: Types, Mechanisms, and Clinical Cardiovascular Consequences. J Am Coll Cardiol. 2017; 69: 841–58.
- Castriotta RJ, Wilde MC, Lai JM, Atanasov S, Masel BE, Kuna ST. Prevalence and Consequences of Sleep Disorders in Traumatic Brain Injury. J Clin Sleep Med. 2007; 3: 349–56.
- Popp RFJ, Fierlbeck AK, Knüttel H, König N, Rupprecht R, Weissert R, et al. Daytime sleepiness versus fatigue in patients with multiple sclerosis: A systematic review on the Epworth sleepiness scale as an assessment tool. Sleep Med Rev. 2017; 32: 95– 108.
- Cacioppo JT, Cacioppo S. Social Relationships and Health: The Toxic Effects of Perceived Social Isolation. Soc Personal Psychol Compass. 2014; 8: 58–72.
- Martin JL, Fiorentino L, Jouldjian S, Josephson KR, Alessi CA. Sleep quality in residents of assisted living facilities: effect on quality of life, functional status, and depression. J Am Geriatr Soc. 2010; 58: 829–36.
- Bloom HG, Ahmed I, Alessi CA, Ancoli‑Israel S, Buysse DJ, Kryger MH, et al. Evidence‑based recommendations for the assessment and management of sleep disorders in older persons. J Am Geriatr Soc. 2009; 57: 761–89.
- Morgenthaler TI, Lee‑Chiong T, Alessi C, Friedman L, Aurora RN, Boehlecke B, et al. Practice parameters for the clinical evaluation and treatment of circadian rhythm sleep disorders: An American Academy of Sleep Medicine report. Sleep. 2007; 30: 1445–59.
- Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989; 28: 193–213.
- Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991; 14: 540–5.
- Carney CE, Buysse DJ, Ancoli‑Israel S, Edinger JD, Krystal AD, Lichstein KL, et al. The consensus sleep diary: standardizing prospective sleep self‑monitoring. Sleep. 2012; 35: 287–302.
- American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events Summary of Updates in Version 2.5. J Clin Sleep Med. 2020.
- Kushida CA, Littner MR, Morgenthaler T, Alessi CA, Bailey D, Coleman Jr. J, et al. Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep. 2005; 28: 499–521.
- Morgenthaler T, Alessi C, Friedman L, Owens J, Kapur V, Boehlecke B, et al. Practice parameters for the use of actigraphy in the assessment of sleep and sleep disorders: an update for 2007. Sleep. 2007; 30: 519–29.
- Marino M, Li Y, Rueschman MN, Winkelman JW, Ellenbogen JM, Solet JM, et al. Measuring Sleep: Accuracy, Sensitivity, and Specificity of Wrist Actigraphy Compared to Polysomnography. Sleep. 2013; 36: 1747–55.
- Irish LA, Kline CE, Gunn HE, Buysse DJ, Hall MH. The role of sleep hygiene in promoting public health: A review of empirical evidence. Sleep Med Rev. 2015; 22: 23–36.
- Erickson AJ, Rodriguez JC, Ravyts SG, Dzierzewski JM, Fung CH, Kelly MR, et al. The effectiveness of cognitive behavioral therapy for insomnia on sleep outcomes in the context of pain among older adult veterans. J Am Geriatr Soc. 2024; 72: 2319–28.
- Morin CM, Colecchi C, Stone J, Sood R, Brink D. Behavioral and pharmacological therapies for late‑life insomnia: A randomized controlled trial. JAMA. 1999; 281: 991–9.
- Riemersma‑van Der Lek RF, Swaab DF, Twisk J, Hol EM, Hoogendijk WJG, Van Someren EJW. Effect of bright light and melatonin on cognitive and noncognitive function in elderly residents of group care facilities: a randomized controlled trial. JAMA. 2008; 299: 2642–55.
- Zisapel N. Circadian rhythm sleep disorders: Pathophysiology and potential approaches to management. CNS Drugs. 2001; 15: 311–28.
- Black DS, O’Reilly GA, Olmstead R, Breen EC, Irwin MR. Mindfulness meditation and improvement in sleep quality and daytime impairment among older adults with sleep disturbances: a randomized clinical trial. JAMA Intern Med. 2015; 175: 494–501.
- Reid KJ, Baron KG, Lu B, Naylor E, Wolfe L, Zee PC. Aerobic exercise improves self‑reported sleep and quality of life in older adults with insomnia. Sleep Med. 2010; 11: 934–40.
- León‑Barriera R, Chaplin MM, Kaur J, Modesto‑Lowe V. Insomnia in older adults: A review of treatment options. Cleve Clin J Med. 2025; 92: 43–50.
- Patel D, Steinberg J, Patel P. Insomnia in the elderly: A review. J Clin Sleep Med. 2018; 14: 1017–24.
- Fick DM, Semla TP, Steinman M, Beizer J, Brandt N, Dombrowski R, et al. American Geriatrics Society 2019 Updated AGS Beers Criteria® for Potentially Inappropriate Medication Use in Older Adults. J Am Geriatr Soc. 2019; 67: 674–94.
- Kumar S, Wong PS, Hasan SS, Kairuz T. The relationship between sleep quality, inappropriate medication use and frailty among older adults in aged care homes in Malaysia. PLOS ONE. 2019; 14: e0224122.
- Hamza SA, Saber HG, Hassan N. Relationship between sleep disturbance and polypharmacy among hospitailzed elderly. Eur J Geriatr Gerontol. 2019; 6: 34–7.
- Epstein LJ, Kristo D, Strollo Jr. PJ, Friedman N, Malhotra A, Patil SP, et al. Clinical guideline for the evaluation, management and long‑term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009; 5: 263–76.
- Kaffenberger TM, Soose RJ. Hypoglossal Nerve Stimulation Therapy for Obstructive Sleep Apnea. Compend Contin Educ Dent Jamesburg NJ. 1995; 44: 332–9.
- Cowie MR, Woehrle H, Wegscheider K, Angermann C, d’Ortho M‑P, Erdmann E, et al. Adaptive Servo‑Ventilation for Central Sleep Apnea in Systolic Heart Failure. N Engl J Med. 2015; 373: 1095–105.
- Fietze I, Blau A, Glos M, Theres H, Baumann G, Penzel T. Bi‑level positive pressure ventilation and adaptive servo ventilation in patients with heart failure and Cheyne‑Stokes respiration. Sleep Med. 2008; 9: 652–9.
- Selim B, Ramar K. Sleep‑Related Breathing Disorders: When CPAP Is Not Enough. Neurotherapeutics. 2021; 18: 81–90.
- Johnson KG, Johnson DC. Bilevel positive airway pressure worsens central apneas during sleep. Chest. 2005; 128.
- Fox H, Oldenburg O, Javaheri S, Ponikowski P, Augostini R, Goldberg LR, et al. Long‑term efficacy and safety of phrenic nerve stimulation for the treatment of central sleep apnea. Sleep. 2019; 42: zsz158.
- Javaheri S, McKane S. Transvenous phrenic nerve stimulation to treat idiopathic central sleep apnea. J Clin Sleep Med. 2020; 16.
- Gagnon J‑F, Postuma RB, Mazza S, Doyon J, Montplaisir J. Rapideye‑movement sleep behaviour disorder and neurodegenerative diseases. Lancet Neurol. 2006; 5: 424–32.
- Howell M, Avidan AY, Foldvary‑Schaefer N, Malkani RG, During EH, Roland JP, et al. Management of REM sleep behavior disorder: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2023; 19: 759–68.
- Winkelman JW, Berkowski JA, DelRosso LM, Koo BB, Scharf MT, Sharon D, et al. Treatment of restless legs syndrome and periodic limb movement disorder: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2025; 21: 137–52.
- Dodson ER, Zee PC. Therapeutics for Circadian Rhythm Sleep Disorders. Sleep Med Clin. 2010; 5: 701–15.
- Zee PC, Vitiello MV. Circadian Rhythm Sleep Disorder: Irregular Sleep Wake Rhythm. Sleep Med Clin. 2009; 4: 213–8.